 The entropy of a closed system, determined. The latter data are fitted by the polynomial C-p = J K-1 mol(-1), which allows extrapolation to high temperatures. The third law provides an absolute reference point for the determination of entropy at any other temperature. 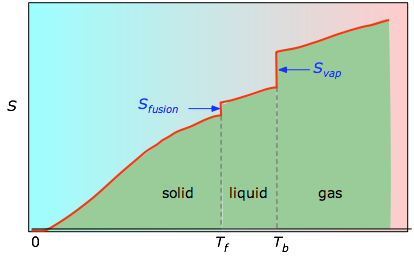
The low-temperature heat-capacity data are compared with the DSC data obtained from 143 K to 775 K and show marginal differences in the common temperature range. 
A relative enthalpy increment of 22,119 J mol(-1) and an absolute entropy value of 127.13 +/- 0.25 J mol(-1) K-1 at 298.15 K are derived from the results. 
No phase transition was observed over this temperature range. The low-temperature heat capacity of epsilon-Mg2PO4OH was measured between 10 and 400 K by adiabatic calorimetry. Heat capacity, on the other hand, can have its absolute value determined experimentally, and it wont depend on a reference value like entropy does. Absolute Entropy, denoted as S, is the measure of disorder or randomness in a substance at an absolute zero temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
